Basic Science Research
![]()
Large Animal/Primate Research
DBC’s Deep Array probes are designed for use in large animals and provide the superb single unit separation and high channel count that has made our silicon probes so popular in awake and behaving non-human primates.
The Hoffman lab is using DBC probes to explore the neural population dynamics underlying learning, memory formation and perception. In a non-human primate model, probes are implanted chronically in deep brain structures (hippocampus, retrosplenial cortex) with multiple 64 and 128 channel implants in a single macaque. Data from up to 256 channels, from multiply-implanted probes, is transmitted wirelessly (Neuralynx Technologies) as subjects behave and move freely in an interactive “tree house.” The DBC team is excited to work with the Hoffman lab in the coming months to produce a custom data analysis pipeline to manage the unprecedented volume of spike and field data using our cloud-based analysis platform.
Kari Hoffman, PhD, Vanderbilt University
![]()
Small Animals/Rodent Research
DBC’s probes give you unprecedented recording density on the smallest possible package for chronic awake and behaving recording in small animals such as mice and zebra finches.
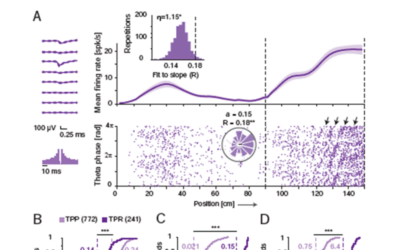
Case Study: High Fidelity Theta Phase Rolling of CA1 Neurons
Single hippocampal cells encode the spatial position of an animal by increasing their firing rates within “place fields”, and by shifting the phase of their spikes to earlier phases of the ongoing theta oscillations (theta phase precession). Whether other forms of spatial phase changes exist in the hippocampus is unknown.
Theta phase precession is a well-known coding scheme in which neurons represent the position of the animal by the timing of their spikes with respect to the phase of ongoing theta oscillations. Here, we show that hippocampal neurons also undergo “theta phase rolling”, a phase change faster and opposite in direction to precession. As the animal advances in space, spikes occur at progressively later phases of consecutive theta cycles. Future studies may reveal whether phase rolling constitutes a novel coding mechanism of space.
Hadas E. Sloin, Amir Levi, Shirly Someck, Lidor Spivak, Eran Stark, Sagol School of Neuroscience and Department of Physiology and Pharmacology, Sackler Faculty of Medicine, Tel Aviv University, Tel Aviv 6997801, Israel

Case Study: Joint Coding of Visual Input and Eye/head Position in V1 of Freely Moving Mice
Visual input to the brain during natural behavior is highly dependent on movements of the eyes, head, and body. Neurons in mouse primary visual cortex (V1) respond to eye and head movements, but how information about eye and head position is integrated with visual processing during free movement is unknown, since visual physiology is generally performed under head-fixation.
The Niell Lab studies how the neural circuits involved in visual processing allow us to complete complex visual tasks, and the effects of development and experience on them. One focus of their work is done in the context of naturalistic behaviors. They have incorporated lightweight, high-density DBC probes to study how eye and body movements affect visual processing in freely-moving mice. This unique and fascinating approach is outlined in the preprint of their publication “Joint coding of visual input and eye/head position in V1 of freely moving mice”, bioRxiv (20222)
Philip R. L. Parker, Elliott T. T.Abe, Emmalyn S. P. Leonard, Dylan M. Martins, Cristopher M. Niell, Institute of Neuroscience and Department of Biology, University of Oregon
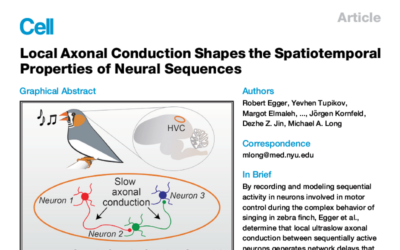
Case Study: Local Axonal Conduction Shapes the Spatiotemporal Properties of Neural Sequences
By recording and modeling sequential activity in neurons involved in motor control during the complex behavior of singing in zebra finch, Egger et al., determine that local ultraslow axonal conduction between sequentially active neurons generates network delays that underlie the pattern of network activity within the circuit.
The acquisition and performance of songs in zebra finches and singing mice is a powerful model for speech and other complex human behaviors. In particular, the way in which coordinated motor movements are planned and executed at the level of neural circuits is poorly understood. The Long Lab is working on this challenging question using a variety of techniques including chronic implantation of high-channel-count DBC probes, resulting in recent publications in Cell (2020) and Neuron (2021). We are also excited to be working with the Long Lab on the translation of these experiments to understanding human speech generation, through our joint collaboration with the University of Iowa.
Michael A. Long, PhD, New York University School of Medicine
Explore all publications using our probes >>
Want to find out more about the latest neurotechnology? Want to discuss your experiments in detail? Need help getting started but not sure where to begin? Need a quote ASAP? Get in touch with our neuroscientist-led sales team…. info@cambridgeneurotech.com
